Can We "Just Stop Oil"?
"There is always an easy solution to every problem - neat, plausible, and wrong"- H. L. Mencken
The slogan - “Just Stop Oil” - is easy to regurgitate. Yet despite pouring national fortunes into the so-called energy transition, net-zero pledges, promises that cheap renewable energy is (supposedly) just around the corner, and now 28 UN Conferences of the Parties (COPs), fossil fuels still dominate.
Based on the 2023 Statistical Review of World Energy, oil, coal, and natural gas supply roughly 82% of global energy, fueling everything from gas power plants to jet aircraft (including the private jets hauling green elites around so that they can lecture the rest of us on using too much fossil fuels). Hydro-electricity provides ~7% of the world’s energy. Wind and solar combined provide ~ 6%, and nuclear ~ 4% (there are other energy sources including bio-fuel and geothermal).
There is nothing wrong with developing new and more efficient ways to extract energy. After-all, it’s the better part of human nature to strive to improve the world. That hunger for improvement helped fuel our march from the caves to the moon and beyond.
But…
…It’s a very, very bad idea to hate on something we all depend on without having a healthy appreciation for why we use it. It’s like standing on ice in the middle of a frozen lake. You might hate the ice and want to get off of the ice, but it’s a terrible idea to smash up the ice around you without understanding that it is the only thing separating you from the watery abyss below.
So before we pledge to chop up our fossil fuel economy, let’s ask a simple question: Why are fossil fuels so damn hard to replace useful?
The Backbone of Life
The secret of why oil and gas is so hard to replace starts with… atoms. No matter how hard they might try, governments can’t re-write the rules of physics and chemistry. Like it or not, carbon is a primary basis for life on earth. Trees, grass, fish, dogs, cats, the food we eat, and all humans are quite literally, carbon-based. If we could see a strand of DNA, carbon and carbon bonds make up large parts of the underlying pieces of DNA, literally helping to hold it together.
Why is carbon so crucial to life? Because of its incredible features, carbon forms the ‘backbone’ of many molecules found in the world around us. Carbon can share 4 electrons with other atoms, enabling various ‘chain,’ ‘ring,’ or ‘branch’-like skeletons for a myriad of molecules.
Because it’s one of nature’s favorite building blocks, it’s just natural that carbon also forms the ‘backbone’ for oil and gas. And like a box of very popular plastic building blocks, there’s a nearly unlimited number of combinations on how they can be put together, ranging from simple to incredibly complex.
Like Trail Mix: A Basket of Products
Readers familiar with the oil business know that oil and gas isn’t a uniform ‘thing.’ Instead, it’s a whole mix and match of different molecules. It’s a bit like a bag of trail mix, with M&Ms, peanuts, almonds, pretzels, etc. And like different trail mixes, crude oil type varies, coming in heavy and light crude, sweet and sour, with just about everything in-between.
To understand the mix, let’s measure the size of a hydrocarbon’s ‘backbone’ by counting carbon atoms. Starting with C1 is natural gas. It just has one carbon atom bonded with 4 hydrogen atoms. Because it’s so small, it can seep through tiny pore spaces in rocks, explaining why some oil and gas wells continue to produce natural gas long after the oil (heavier molecules) production has dropped off. Incidentally, based on energy content, natural gas is incredibly cheap compared to oil. By weight, it is extremely energy dense. On the flip side, because it is so small and light you need to keep it under extreme pressure or very cold, making its above ground storage significantly more complex than oil storage.
You can quickly move your way up the scale. C2 is ethane. Ever BBQ with a gas grill? You probably used propane (C3) made up of, no surprise, 3 carbon and 8 hydrogen atoms. C4 is butane found in lighters.
Around C5, it becomes a liquid, bordering between oil and gas. Oil that ‘flows’ at 60 degrees F go up to around C20 (20 carbon atoms per molecule). After that, oil becomes a semi-solid and eventually a solid.
As you get longer molecules, the ratio of hydrogen and carbon changes, impacting the different fuels’ behavior. That’s not the whole story but it gets us in the ballpark.
Sorting It All Out.
Where you drill for oil and gas can impact the mix that you get out of the ground. But, nature isn’t perfectly organized and you can’t typically just find a pure source of one type of molecule.
Let’s just focus here on oil, assuming the natural gas stream is separated from oil at well-site (as most of it often is).
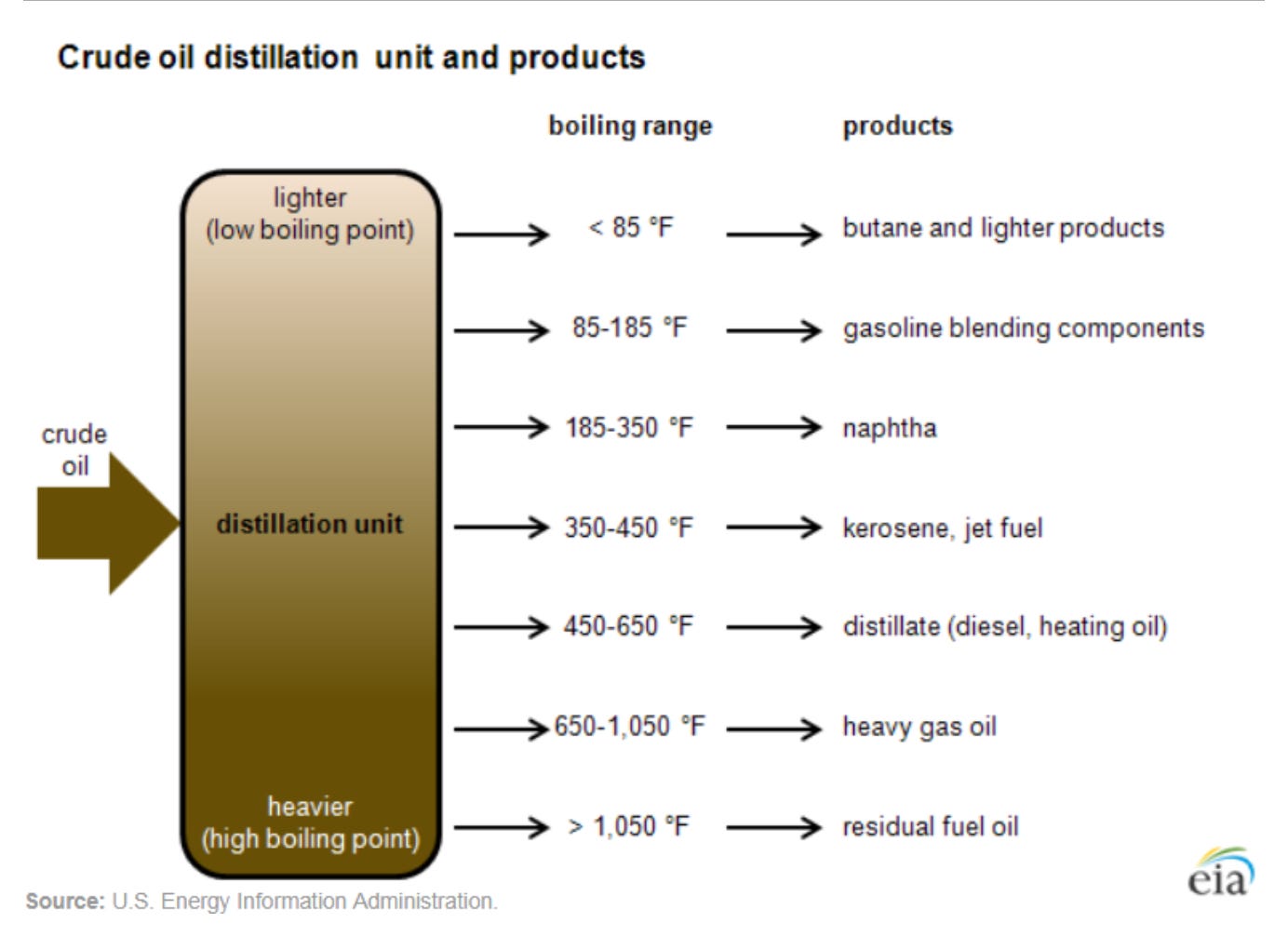
Unrefined crude isn’t very useful by itself. However, that mix of molecules becomes extremely useful once it’s refined. Fortunately for us, heat can sort the different molecules by their weight (basically, by how many carbon atoms they have).
However, you (typically) can’t turn all of 1 barrel of oil into just 1 barrel’s worth of jet fuel or just 1 barrel’s worth of gasoline. Each product has a range of molecules. They can overlap and an oil refiner can change the mix of gasoline, jet fuel, and diesel, but only up to a certain point.
For example, gasoline is made from a blend of molecules ranging from c4 to c12. Jet fuel is made from c8 to c16. And diesel is made with molecules ranging from 8 to 23 carbon atoms. (But those are ranges, and not the ideal mix per se.)
That means there’s only so much jet fuel or diesel that you can get out of a barrel- the rest are ‘leftovers.’ It’s a lot like buying trail mix just for the M&Ms. After you eat them, there’s still quite a bit leftover.
Waste Not, Want Not.
That’s exactly what the oil and gas business did - find uses for just about every last molecule in oil and gas. Today, uses for oil and gas fall into 4 major ‘buckets.’ And parts of the same barrel of oil can end up in multiple buckets. The ‘4 buckets’ are:
Fuels: think of jet-fueled aircraft, diesel trucks, propane grain dryers, and yes gasoline cars. Bio-fuels and renewable fuels end up looking a lot like the petroleum products: they’re a round-about way to get what we pretty much already have from oil and gas.
Pharmaceuticals: is that a surprise? Our life is carbon based and it should be not shock that hydrogen and carbon are used as ingredients in medications.
Fertilizer: It is essential for food production to feed 8 billion souls. Literally, we couldn’t feed the world without fertilizers made directly with natural gas or coal ingredients. Even as the greens try to reinvent fertilizer production, the end product looks similar to the fertilizer made with natural gas. The difference is that it is generally a lot more complex to make the ‘green’ fertilizer than it is to simply use natural gas to make it.
Chemical feedstocks: over 6000 common products- ranging from makeup to smartphones, tires to asphalt, and synthetic clothes to glues- are made with oil and gas byproducts.
The world is highly interdependent- food, fuel, feedstocks for medicine and modern products are all closely linked. And this is what many of the green wannabe central planners miss - you can’t waive a magic wand and simply only change the thing you want to change. That’s one of the challenges of the so called ‘energy transition.’ How do you reduce demand for an entire basket of products that come from oil and gas?
Here’s how it could play out. Under the guise of reducing carbon, the all-knowing central planners might drive down gasoline demand in one part of the world (in the west for as long as we faithfully believe in the UN climate targets). But as long as we still need diesel, jet fuel, and chemical feedstocks, what are we going to do with the ‘surplus’ gasoline? Most likely, we’ll find another use for it or just sell it to other parts of the world hungry for cheap energy.
Nature’s Battery
But it gets better. In part because of carbon’s unique structure (4 covalent bonds), hydrocarbons are an excellent store of energy, forming one of nature’s chemical batteries. When those hydro-carbon bonds get broken and reformed, they release enormous amounts of energy. Just a tiny bit of energy can start a chain reaction, allowing you to turn on a gas stove and heat up dinner.
Just how much energy is packed into oil and gas depends on the end product. But let’s compare diesel’s energy density to a Tesla 4680 battery. By weight, diesel packs ~ 36x as much energy. Yes, a Tesla is better at converting energy from a battery into electricity to drive the car than a diesel truck is at converting fuel into work. But even if we assume a Tesla is 100% efficient and a diesel truck is only 40% efficient, diesel fuel packs ~14x the energy by weight, after accounting for engine efficiency. And we’re assuming the Tesla battery stores the ideal amount of energy, regardless of the weather.
But…What about water? Can’t we do the same with H2O? No, unfortunately, oxygen has different bonding characteristics so it doesn’t release energy in the same way as hydrocarbons. In fact, it takes more energy to split the water (into hydrogen and oxygen) than it releases. Hydrocarbons release far more energy than it takes to start splitting them up. So, they’re a net energy source. In contrast, hydrogen is a net energy drain, taking more energy to ‘make’ it than it provides.
Now we’re starting to really paint a pictures of why hydrocarbons are so useful. To close, let’s look at history to see how the carbon backbone, the basket effect, and energy storage came together to help make the modern world so dependent on oil and gas.
Replacing Whale Oil
Before we used petroleum oil, we had whale oil. Whales were harvested for oil used for lighting, making soap, lubricating machinery, etc. But, whale oil had major downsides. As the tale Moby Dick highlighted, chasing down and harpooning massive mammals is a hazardous occupation.
(As a side note, the first mate in that novel is Mr Starbuck. His name inspired in part, the name for the coffee giant, Starbucks.)
Whale oil is a type of ‘bio’ oil and the rate of replacement is key. When demand for the oil outstripped the whales’ ability to reproduce, the whale population declined. But, despite the overhunting of whales, people still needed oil for lighting. Once people have tasted a certain standard of living, it’s really hard to un-taste it. So, humans searched for a substitute. And, since whale oil is made mostly of hydrogen and carbon, it’s no surprise that humans found other sources, mostly from hydrogen and carbon.
Early oil speculators were drawn to western Pennsylvania where ‘seep oil’ naturally flowed to the surface in the area aptly named Oil Creek. Hamilton McClintock - a farmer in the area - even built a wooden structure to trap oil on the surface of the creek. (And just to touch on this, oil and gas is abundant in many parts of the world. Plus oil is fairly easy to transport and store, making it all the more appealing.)
But back to the story…
A group of investors, Eveleth and Bissell, hired a Yale chemistry Professor, Benjamin Silliman to study what could be done with the ‘rock oil’ as it was called. Silliman used fractional distillation to ‘sort out’ the parts of the oil. He found that the distilled oil was very good as a lighting fuel and published his report in 1855.
Within a few years, Edwin Drake and “Uncle” Billy Smith drilled the first successful oil well in 1859. Initially, it wasn’t easy going. Uncle Billy accidentally burned down the derrek and house. And then there was the ground water: it flooded the well, causing it to collapse around 16 feet below the surface. Eventually, Drake drove a conductor pipe into the ground first. The pipe protected the well from the groundwater and collapse. That did the trick, enabling them to drill past the water table to strike oil, setting off an oil boom in western PA.
From Garbage to Gold
Initially, oil was mainly used for kerosene lighting. Much like buying trail mix for the M&Ms, early refiners didn’t know what to do with an oil ‘byproduct’ we now know as gasoline. At that time, it was considered waste and discarded.
By the turn of the 19th to 20th century, automobiles were still a novelty and the majority of cars were either steam powered or electric vehicles. The adoption of the gasoline engine- with its power density - was driven in part by its ability to use waste gasoline.
The gasoline story has parallels with the rest of oil and natural gas. Driven by a desire to not waste a drop, we developed countless byproducts now indispensable for food production, industry, power generation, transportation and feedstocks for over 6000 products.
Just for fun, the next time you see a ‘just stop oil’ protest on TV or in person, why not play a little game? Try counting how many oil and gas based products the protesters are either using or wearing. Chances are, you’ll end up with a very long list! For reference, energy.gov provides a list of some of the common products made from oil and gas.
AsRobert Bryce says “If oil didn’t exist, we’d have to invent it.” Whale oil is made mostly of hydrogen and carbon, and it was replaced with petroleum, made up with similar atoms. Even today as we race to invent ways to get rid of oil and gas, we’re having to invent or find new ways to create chemical compounds that mimic…oil and gas. And that’s a true testament to how important petroleum is: when we’re trying to get rid of it, we end up having to re-invent oil in an attempt to replace…oil. It’s a bit like trying to re-invent the wheel - that new wheel probably still looks round.
As always, thanks for reading!






Excellent article
One point of clarification: when we talk of breaking molecules in the report, we’re referring to the net of breaking and forming new molecules. Energy is absorbed when breaking and releasing when forming new molecules. The net is key.